Magnesium reagents
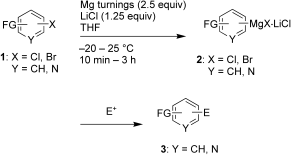
The simple LiCl-mediated insertion of magnesium into aryl chlorides and bromides at moderate temperatures leads to fuctionalized organomagnesium reagents.
An unprecedented range of functionalized groups may be present in the substrates (e.g. CN, CO2R, OTs, OBoc; Ts=p-toluenesulfonyl, Boc=tert-butylcarbonyloxy).
- Piller, Fabian M.; Appukkuttan, Prasad; Gavryushin, Andrei; Helm, Matthew; Knochel, Paul. Convenient Preparation of Polyfunctional Aryl Magnesium Reagents by a Direct Magnesium Insertion in the Presence of LiCl. Angew. Chem. Int. Ed., 2008, 47, 6802-6806.
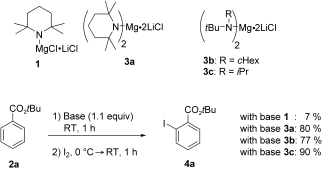
Because some moderately activated arenes such as tert-butyl benzoate or benzonitrile gave unsatisfactory results. Therefore, we have developed a new class of mixed Li/Mg bases: magnesium bisamides complexed with lithium chloride, (R2N)2Mg 2 LiCl.
2 LiCl.
- Clososki, Giuliano C.; Rohbogner, Christoph J.; Knochel, Paul. Direct Magnesiation of Polyfunctionalized Arenes and Heteroarenes Using (tmp)2Mg2 LiCl. Angew. Chem. Int. Ed., 2007, 46, 7861-7684.
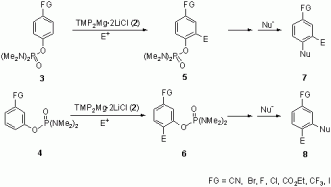
The DMG N,N,N',N'-tetramethylphosphorodiamidate ((Me2N)2P(O)-O-) is a very strong directing group for magnesiation and that it may overrule the effects of other substituents present on the aromatic substrate.
This would allow new types of functionalization such as formal meta or para functionalization.
- Rohbogner, Christoph J.; Clososki, Giuliano C.; Knochel, Paul. A General Method for meta and para Functionalization of Arenes Using TMP2Mg
 2 LiCl. Angew. Chem. Int. Ed., 2008, 47, 1503-1507.
2 LiCl. Angew. Chem. Int. Ed., 2008, 47, 1503-1507.
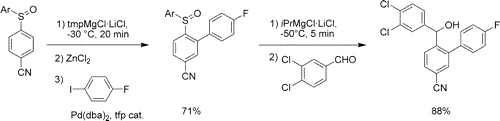
The aryl sulfoxide moiety (ArSO) allows an expedient two-step meta-, para-difunctionalization of readily available diaryl sulfoxides. In the first step, the sulfoxide plays the role of a directing metalation group. In the second step, triggered by i-PrMgCl·LiCl, it becomes a leaving group and undergoes a regioselective sulfoxide−magnesium exchange.
- Rauhut, Christian B.; Melzig, Laurin; Knochel, Paul. Meta- and Para-Difunctionalization of Arenes via a Sulfoxide-Magnesium Exchange Reaction. Org. Lett., 2008, 10, 3891-3894.
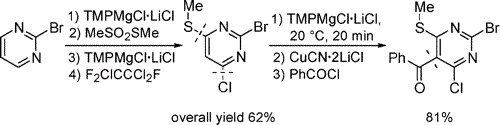
Successive regio- and chemoselective magnesiations of pyrimidines using TMPMgCl·LiCl furnish, after trapping with various electrophiles,
highly functionalized derivatives in good to excellent yields. Applications to the synthesis of antiviral and anti-inflammatory agents such as
p38 and sPLA2 kinase inhibitors are reported.
- Mosrin, Marc; Knochel, Paul. Regio- and Chemoselective Multiple Functionalization of Pyrimidine Derivatives by Selective Magnesiations using TMPMgCl•LiCl. Org. Lett., 2008, 10, 2497-2500.




